EMFACE – Fewer Wrinkles and Natural Lift Without Needles
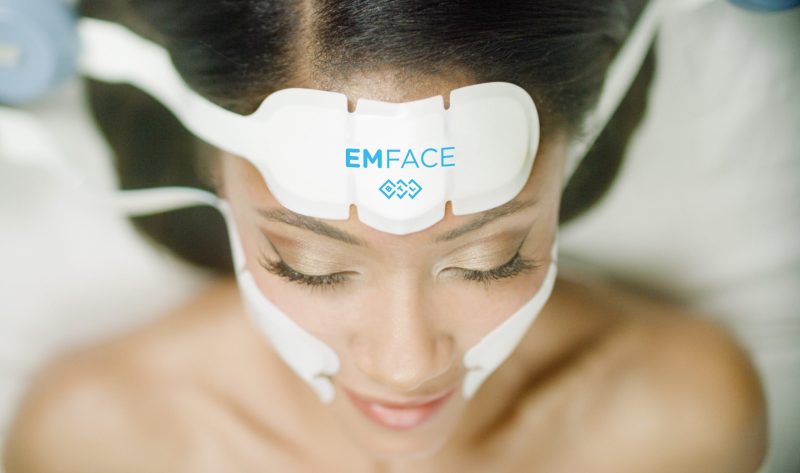
What is EMFACE?
EMFACE is the first treatment that affects the structure of your face from within – naturally without needles. EMFACE treatment is the first method that treats both the skin and muscle tissue of the face simultaneously, using a patented combination of Synchronized RF (Radio Frequency) therapy and HIFES (High-Intensity Focused Electromagnetic Stimulation) for facial muscle stimulation.
Synchronized RF heats the dermis to stimulate collagen and elastin production, while HIFES selectively contracts facial muscles. This results in more effective reduction of wrinkles and fine lines as well as an overall lift of the face. The efficacy and safety of EMFACE has been confirmed by clinical studies and it has been approved by the U.S. Food and Drug Administration (FDA). Clinical studies showed an average reduction of 37% in wrinkles, 30% increase in muscle growth, and a 23% increase in lift.
EMFACE restores a strong and healthy foundation to the superficial musculoaponeurotic system (SMAS), enabling facial contouring with other methods if desired. Best of all, EMFACE treats the entire face in just 20 minutes (a series of 4 treatments once a week is recommended).



High-Intensity Facial Electromagnetic Stimulation (HIFES Technology)
Benefits
- HIFES produces 75,000 facial muscle contractions in each 20-minute session. The targeted muscles include the frontalis muscles, which lift the eyebrows, and the zygomaticus major, minor, and risorius muscles of the cheeks, which lift the corners of the mouth and elevate the midface.
- These rapid contractions trigger cellular changes that increase muscle volume, density, and tone, providing the face with more structural support.
Synchronized Radiofrequency Energy (Sync RF)
- Synchronized RF (radiofrequency) energy penetrates the dermis and heats the tissue to 40-42 degrees Celsius within minutes during the treatment. This temperature is sufficient to stimulate new collagen and elastin growth without affecting the facial fat tissue.
- Collagen and elastin are structural proteins that give the skin its elasticity, strength, and resilience. We produce less of them as we age, leading to signs of facial aging.
- Stimulating new collagen and elastin growth in the dermis results over time in firmer, smoother, and more elastic skin.
Does EMFACE Work?
Clinical studies provided by BTL show that EMFACE works: A series of four treatments results in a 30% improvement in muscle firmness, a 23% lifting effect (especially in the brow and cheek areas), and a 37% reduction in wrinkles, with a patient satisfaction rate of 95%.
These studies also showed that collagen levels increased by 26% and elastin levels rose by 129%. Collagen remodeling and muscle fiber growth take time, so optimal results are visible three months after your last treatment.
It is important to have realistic expectations about what any non-invasive facial rejuvenation treatment can achieve. EMFACE does not replace surgical facelifts, which lift deeper muscles and skin by removing excess tissue. But it can be a good option for patients seeking a natural-looking improvement without the risks, recovery time, or scars associated with surgical procedures.
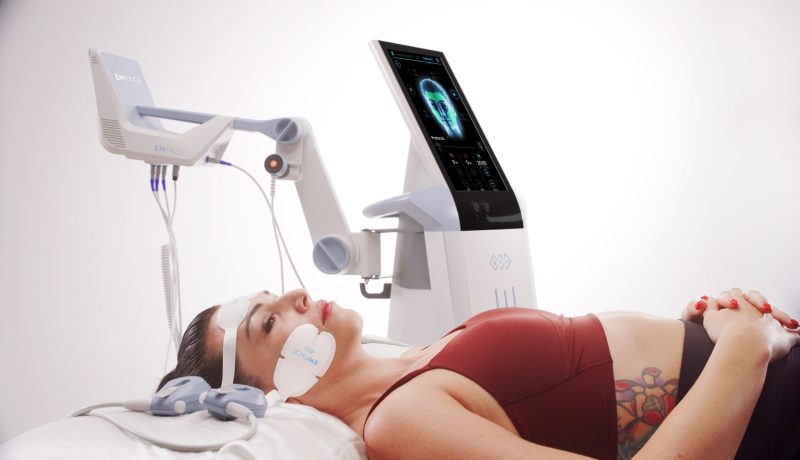
What are the pros and cons of EMFACE?
Pros
- This non-invasive treatment requires no recovery time. After the treatment, you can wear makeup, exercise, or return to work without worry.
- It is suitable for all skin types and tones.
- The 20-minute treatment is not painful, even without using numbing cream.
- The results look very natural. The treatment restores patients to their natural baseline. EMFACE is an excellent option for patients who are hesitant about looking too “overdone” with neurotoxins and fillers.
- EMFACE restores a strong and healthy foundation – to the superficial musculoaponeurotic system (SMAS), which allows for facial sculpting with other methods as well, if desired.
Cons
- EMFACE is not a one-time procedure: your treatment plan will likely include four weekly sessions followed by occasional maintenance treatments.
- Although you will notice and feel some of the benefits of EMFACE after your first treatments, you will need to wait about three months after your last session to see the optimal results. This is the time it takes for new collagen, elastin, and muscle fibers to develop.
- Results last about a year, so you will need to return for maintenance treatments if you want to maintain them.
- The treatment cannot address the neck area.
- This treatment is not recommended for individuals with facial implants or any metal in the face (piercings should be removed from the treated area), nor is it recommended for those with pacemakers, active inflammatory conditions, or tattoos in the treatment area.
Who is EMFACE best suited for?
The advantage of EMFACE is that it suits most patients. There are no restrictions regarding skin type or tone, so almost anyone who wants to lift their face, tighten the skin, and smooth wrinkles is a good candidate for EMFACE.
However, there are a few exceptions. People with implantable electronic devices, such as pacemakers, or metal implants that may interfere with electromagnetic waves near the treatment area are not good candidates.
Patients with advanced signs of aging, such as significant skin and muscle laxity, may benefit more from a facelift surgery.
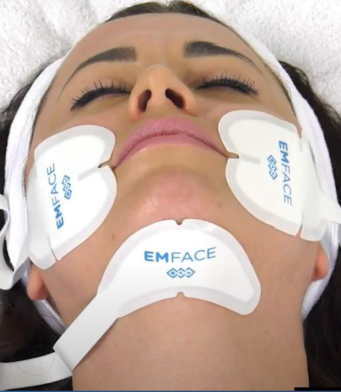
What to Expect During an EMFACE Treatment?
Typical Treatment Process:
- You relax on the treatment table while the practitioner thoroughly cleanses your face.
- EMFACE electrodes are placed on your forehead and cheeks, and the treatment begins.
- During the treatment, you will feel rapid muscle contractions and a warming sensation. This combination can cause a tingling or unusual feeling, but it is not painful. You may notice involuntary facial expressions or movements, but this is completely normal.
- After approximately 20 minutes, you can return to your normal daily routine. The skin may be slightly red, but otherwise, no one will likely notice that you just had a treatment.
How Soon Will You See Results?
Typically, patients notice improvements after completing a series of four treatments. Some may see results even sooner, such as a lift in the eyebrows and jawline, a more defined jawline, and improved skin appearance after just one treatment.
The results experienced by patients can vary from subtle enhancements to more dramatic changes. It's important to note that the results will continue to improve for three months following the last treatment.
How Long Do EMFACE Results Last?
According to clinical studies and feedback from providers, EMFACE results can last up to a year.
You can support long-lasting results by maintaining a high-quality skincare routine, including protecting your skin with sunscreen that has at least SPF 50+. After one year, one maintenance treatment per year is recommended, but this can vary depending on the patient's skin condition and age.
Is EMFACE Safe? Does It Cause Any Side Effects?
EMFACE has an excellent safety profile, and no long-term adverse effects have been reported. Potential side effects are mild.
In clinical practice, EMFACE treatments have been found to be very well tolerated, with patients reporting only minor side effects, such as temporary dry skin, redness, or mild headaches. Anecdotally, there have also been reports of improvements in chronic headaches, neck pain, and heavy eyelids.
Frequently Asked Questions
EMFACE is suitable for anyone looking for fewer wrinkles and more lift. However, there are a few exceptions. It is not suitable for people with implantable electronic devices, such as a pacemaker or metal implants that may interfere with electromagnetic waves near the treatment area.
Contact Information
MESQ®
Subsidiary of DoctusPlus Oy
Company
DoctusPlus Oy
Business ID
3154088-6
Visit Address
Itämerenkatu 11-13 F, 00180 Helsinki
Phone
Opening Hours
Mon – Fri 11:00 – 19:00